John C. Palumbo and Barry Tickes
(PDF
version, 70KB)
Introduction
A number of insects can cause serious problems in broccoli and cauliflower
grown for seed. Sucking insects such as aphids and false chinch bugs can
be serious pests later in the season during reproductive growth periods
when plants are actively being pollinated and producing seed. These pests
injure the crops primarily by feeding on the developing seed. Aphids feeding
in blooms and seed pods are particularly easy to kill with most conventional
OP and carbamate insecticides because they are directly exposed to foliar
sprays. However, aphid control becomes a problem during pollination when
the crop is in bloom and bees are actively foraging in the field. Insecticides
presently registered for use in seed crops are considered highly toxic
to bees. Thus, to avoid killing bees, pollination often must be interrupted.
The colonies are either temporarily removed from the field, or applications
must be made at night. In either case, several days can pass before the
bees resume normal foraging activity. Repeated applications to control
aphid populations may cause a significant disruption in pollination and
reduced seed yield and quality.
A number of selective aphicides are being developed that provide selective aphid control, with little or no toxicity or repellency to honey bees. Fulfill® (pymetrozine, Novartis) is presently labeled on leafy vegetables and has an SLN registration for Brassica seed crops in Arizona (AZ-000004). Our experiences in lettuce suggests that the product can provide 10-14 residual control of aphids when applications are initiated when aphids first begin to colonize on foliage. Other promising candidates (not labeled) Aphistar® (triazimate, Rohm&Haas) and Pirimor® (pirimicarb, Zeneca) have excellent contact and systemic aphid activity on a number of crops. Similarly, studies in lettuce have shown that these two compounds provided about 14-21 d residual aphid control. Unfortunately, both products are carbamates, and their future status is not known. Another promising product under consideration is Assail® (acetamiprid, Ceragri), which is also safe to pollinators. The compound is a neonicitinoid, similar in activity to Admire® and the product was recently granted a registration for use in broccoli and cauliflower in California. In previous field trials, it has shown good control of green peach aphid on lettuce and broccoli. Selectivity can also be achieved by applying the pesticides to the plant without exposing pollinators to the toxicant. Platinum® (thiamethoxam, Syngenta), also a neonicotinoid, has recently been labeled on melons for whitefly control with labels pending on leafy vegetables and Brassica crops for aphids. It is applied in-furrow at planting and has similar activity as Admire. The primary difference between the two products is that Platinum is very mobile in the soil and moves readily with furrow irrigation. Trials in head lettuce and melons have shown that residual aphid control is actually better when the product is applied at side dress rather than at planting. This approach extends residual control closer to harvest. It may be possible to use this approach in Brassica seed crops by side-dressing the product at layby, thus providing residual control at the time aphids begin to colonize in the spring. Given that the products will soon be labeled on cole crops, evaluating this approach may result in an effective management tool. We completed a replicated study in 2003. Note: Admire is not labeled for use on seed crops.
Because the value of these compounds for aphid control in Brassica seed crops grown in Arizona needs to be determined, we conducted studies to measure aphid control with these new experimental compounds under non-blooming (early aphid development) and blooming (heavy aphid pressure) conditions. We also studied aphid population dynamics by identifying and quantifying the aphid species complex on a broccoli seed crop and determined their distribution on plants during the pre-bloom growth period. Ultimately, our goal is to gather data to assist Arizona seed growers and PCA’s in making management decisions and to support registrations for new insecticides.
Materials and Methods
2001 Study: The study was conducted in a 5 acre commercial
cauliflower seed field in Yuma, Arizona. Male transplants were established
in the field on single row, 42 inch beds on 20 September, 2000 and female
transplants were established on 30 September. Plants were furrowed irrigated
throughout the trial. Large plots were established in the field by equally
dividing the field into plots 10 beds wide by 600 ft long. For the insecticide
study, untreated plots were established within randomly selected plots
and were 10 beds wide by 75 ft long. Equal numbers of male and female
plants were distributed equally on alternating beds within each plot.
Each treatment was replicated three times in a randomized complete block
design.
Foliar applications were made with a commercial Melroe sprayer (Dune Co.,
Yuma, AZ) that delivered a directed spray at 25 gpa and 40 psi through
2 nozzles per bed. The first application was made on March 8th when female
plants were in full bloom. Pirimor 50 DF @ 0.5 lb ai/acre, Fulfill 50
WG @ 0.17 lb ai/acre, and Aphistar 50 W @ 0.124 lb ai/acre were applied
at about 1800 hr. A 2nd application was made in a similar manner on March
17th. However, Fulfill was not reapplied, and those plots were over-sprayed
with Pirimor (see table below). Plants were still blooming and bees were
actively working. A crop oil concentrate was added to each treatment at
3 oz/ac.
| Spray TMT #1 | Spray TMT #2 | Rate /ac. |
|---|---|---|
| 1. Pirimor 50DF | Pirimor 50 DF | 8 oz |
| 2. Aphistar 50W | Aphistar 50W | 4 oz |
| 3. Fulfill 50WG | Pirimor 50 DF | 4.5 oz |
| 4. Untreated check | – | – |
Aphid populations were assessed by visually examining plant leaves and
terminals and recording the number of wingless, colonizing aphids. Two
sampling units were used in this study; a single frame leaf from individual
plants, and infested seed pod extensions. Prior to the first application
(pre-treatment samples), 40 female and 40 male plants were randomly selected
across the field and examined. On each sampling date 5 young (a fully
expanded leaf about 4-5th nodes from the terminal) and 5 old (3-4th leaf
position from the base of the plant) leaves per plot were examined and
the number of aphids per leaf were counted. Once bloom began and plants
began to set seed, aphid colonies were visually examined on seed pod extensions.
The number of seed pods (extensions) infested with aphids was recorded.
The number of aphids on each pod was estimated using two methods. When
only a few infested pods were found (ie., treated plots), the number of
aphids on each pod was counted in the field When a large number of pods
was found infested with high aphid densities (ie., untreated plots), 5-8
representative pods from each plot were collected, taken into the laboratory,
and the number of aphid per pod was estimated by direct counts. Treatment
means were analyzed using a 1-way ANOVA and means separated by a protected
LSD (P<0.05).
2003 Study: The study was conducted in a 2-acre broccoli ‘Marathon’ seed field at the Yuma, Arizona. Male and female transplants were established in the field on single row, 42 inch beds on 4 and 25 October, respectively. Plants were furrowed irrigated throughout the trial. Plots were established in the field by equally dividing the field into plots 8 beds wide by 80 ft long. Equal numbers of male and female plants were distributed equally on alternating beds within each plot. Each treatment was replicated three times in a randomized complete block design. For insecticide trial the following 8 treatment combination were evaluated.
| Spray #1 (Feb 18) |
Rate/ac | Spray #2 (Mar 22) | Rate/ac | Spray #3 (Apr 4) |
Rate/ac | |||
|---|---|---|---|---|---|---|---|---|
| 1 | Pirimor 50DF | 3 oz / acre |
Pirimor | 3 oz |
Pirimor | 3 oz |
||
| 2 | Fulfill 50WG | 2.75 oz |
Fulfill | 2.75 oz |
Fulfill | 2.75 oz |
||
| 3 | Assail 70W | 1.7 oz |
Assail | 1.7 oz |
Assail | 1.7 oz |
||
| 4 | MSR + Capture | 2 pts + 6 oz |
Capture | 6 oz |
Capture | 3 oz |
||
| 5 | Platinum-sidedress | 8 oz |
Capture | 6 oz |
||||
| 6 | Admire--sidedress | 24 oz |
Capture | 6 oz |
||||
| 7 | Admire--sidedress | 24 oz |
||||||
| 8 | Untreated | - |
Untreated | - |
Untreated | - |
Several insecticide treatments were applied in this trial. A single Platinum (8 oz/ac) and two Admire (24 oz/ac) treatments were side-dressed on the shoulder of beds, 2-3 inches below the soil in 30 gpa final dilution on Nov 30. Foliar applications were made with a Melroe sprayer (UA YAC) that delivered a directed spray at 25 gpa and 40 psi through 3 nozzles per bed. The first application was made on Feb 18th at 1800 hr. (pre-bloom). A second application was made on March 22nd . Plants were blooming and bees were actively working the field. A third application was made post-bloom on Apr 4 after the bees were removed.
Aphid populations were assessed similar to the 2001 study by visually examining young and old leaves from both male and female plants prior to the first spray application, and seed pod extensions during seed production. Beginning in mid-November, sampling was initiated to determine the abundance and location of aphid species during the pre-blooming growth period. We randomly sampled 80 male plants and 80 female plants about every 14 days from the 2-acre block. From each plant we selected an older leaf from the frame and a younger leaf from the terminal area of the plant. All aphids on each leaf were identified to species and counted. Following the spray applications, the number of seed pods (extensions) infested with aphids was recorded. The number of aphids on each pod was estimated on March 30 by randomly selecting 3 light-moderate and 3 heavy infested pods from each plot and counting the total number of aphids. Treatment means were analyzed using a 1-way ANOVA and means separated by a protected LSD (P<0.05).
Results and Discussion
2001 Study: During the pre-bloom growth period (January-March), our plant counts revealed that two distinct aphid populations were distributed within the cauliflower plants. The frame leaves were largely infested with green peach aphids (GPA >90% ) with a small proportion of cabbage aphid and turnip aphid present. Conversely, as plants began to extend and flower production began, the plant terminals and seed pods were predominately infested with cabbage aphids (CA; > 90%) and a small percentage of GPA (Table 1).
The female plants were in full bloom when the 1st application was made. Tables 1 and 2 show the subsequent changes in aphid populations at 3 and 7 DAT, respectively. Both the Pirimor and Aphistar provide excellent knockdown of established CA on plant terminals and seed pods and resulted in significantly fewer infested seed pods compared with the untreated control. The Fullfill spray provided good control of the GPA found on the frame leaves, but provided marginal knockdown of CA on plant terminals and seed pods. Although aphid numbers were not statistically different among the insecticide treatments, it was clear that the Fulfill treatment allowed a much higher number of aphids to remain in seed pods. Furthermore, the aphid numbers increased significantly in the Fulfill treatment from 3 to 7 days after treatment. Consequently, we applied a second application of Pirimor to the Fulfill plots.
Following the second application (Tables 3 and
4), the Aphistar and Pirimor applications significantly reduced aphid
numbers and infested pods for 28 days following the application. No damage
to the seed pods in treated plots was noted. Aphid numbers in the untreated
plots declined steadily during the 28 day period but this was due to the
large number of lady beetles that migrated into the plots. Unfortunately
most of the seed pods in the untreated plots were seriously damaged due
to excessive aphid feeding.
The results of this study suggested that several selective insecticides,
in particular Pirimor, have good activity on aphids and also offer bee
safety,. None of the products that we applied appeared to affect the foraging
honey bees. Furthermore, we did not observe dead bee in the plots. However,
Pirimor is an carbamate insecticide and may not be registered for seed
crop uses for years if even at all. Similarly, Aphistar has good bee safety
and aphid activity, but registration has not been pursued by the manufacturer,
and will not likely be registered in the future, as it is also an carbamate.
Fulfill does provide bee safety, but did not provide what we would consider
acceptable control in the 2001 study. Unfortunately we were looking forward
to retesting these effects in 2002, but we unable to because our plots
were oversprayed from an adjacent field The compound that we were most
interested in looking at in 2002 was Assail. This compound is a neonicotinoid
(same chemistry as Admire), and has been shown to provide good bee safety.
It has excellent activity against green peach aphid in head lettuce and
cole crops, and was thought to provide excellent control of cabbage aphid
on exposed seed pods.
2003 Study: Similar to 2001, results from our pre-bloom
sampling surveys showed that the primary aphid species found were cabbage
aphids, turnip aphids, and green peach aphids. Geeen peach aphids tended
to colonize plants slightly earlier and were found to primarily on the
older frame leaves low on the plant (Table 6). In
most cases, male and female plants were colonized to the same extent.
Their numbers peaked in late January, and were found on seed pods at very
low densities. Cabbage and turnip aphids behaved similarly, and were combined
for this summary. There appeared to be no clear preference between their
colonization on older and younger leaves. Similarly, they appeared to
colonize males and females equally. During the bloom period (March-April),
the population was almost exclusively cabbage aphid feeding on seed pods
and extensions. Their numbers were higher than green peach aphid, and
rapidly colonized seed pods and extensions once pollination was completed
and bloom had dropped. Cabbage aphid was the primary species (>90%)
found feeding on developing seed pods in this test. Based on both years
results, sampling for aphids on seed crops should begin a week or so prior
to bloom. Scouting should focus on younger leaves from the terminal area.
Once blooming begins, sampling should focus almost exclusively on newly
developing seed pod extensions.
The first spray application made to the foliage of pre-blooming plants
did not apper to significantly reduce population densities (Table
7). This may in part due to lower densities at this time, but also
the tendency for the aphids to move into the developing seed pod extenstions
(Fig 1). Cabbage aphids were the primary species present
on seed pods, and were easily knocked down by contact materials Capture
and MSR. Estimates of actual densities per seed pod extension showed that
contact activity varied by compound. Perhaps most impressive was the estimated
population levels of cabbage aphids on untreated plants (Table
8 and 9). These high levels were sustained in
the check plots for the duration of the plot and resulted in significant
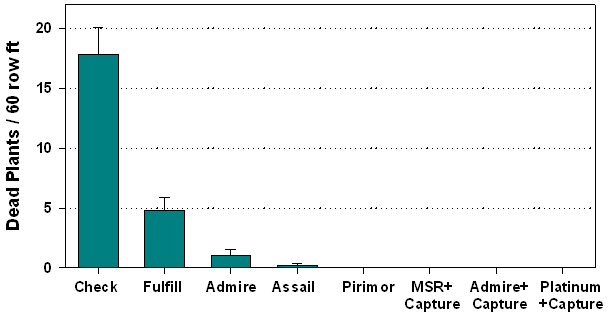
mortality once seed set was complete (Fig 2), and
ultimately resulted in 100% mortality of seed pod extensions in the untreated
check due to aphid feeding.
Of the bee safe products, Pirimor provided the most consistent residual aphid control similar to 2001. This was achieved with 3 applications at a 3 oz rate. Both Assail or Fulfill worked well on foliage, but did not provide comparable control on seed pods. Given Fulfill’s weakness as a contact material on seed crops, it should not be recommended for cabbage aphid control on broccoli seed crops. Although Assail performed better, it needs to be further evaluated before recommendations can be made. Both the side-dress applications of Platinum and Admire provided good aphid suppression prior to seed pod extension. A single Capture application following bloom rapidly controlled colonizing aphids in these plots. The Capture spray will likely provide some control of false chinch bugs on maturing plants (False chinch bugs were not present in the test). The Admire only treatment did not provide adequate protection from aphids during seed production.
In general, the pre-bloom application did not appear to prevent aphids from quickly colonizing seed following extension and bloom (Table 7, Fig 1). In years where aphids number are lighter (such as 2001), a pre-bloom application may significantly suppress colonization.. As noted above, aphid densities were extremely high in the untreated check, and to a lesser extent in the Fulfill plots. The aphids had a direct impact on plant mortality. We had intended to measure seed quality in the plots, but seed yields were not taken due to heavy late season losses to Sclerotinia and bird damage in all plots.
Acknowledgments
Funding for this research was provided by the Seed Trade Association of Arizona. We would like to thank Sakata Seed Co., Yuma, AZ for providing the transplants at no-cost for the 2003 study. We would also like to thank Dune Co. of Yuma for making the spray application at no-cost in the 2001 study. We also acknowledge the following personnel at the Yuma Agricultural Center, Clay Mullis, Andreas Amaya, Luis Ledesma, Leonardo Chavez, Javier Ruiz and Gerardo Villegas for their assistance in completing this project.
| Table 1. Within plant distribution of aphid species in pre-blooming cauliflower, Yuma Mesa, Spring 2001. | |||||||||
Mean aphids
/ leaf |
|||||||||
Cabbage/Turnip
Aphid |
Green Peach
Aphid |
||||||||
| Sample date | Leaf a | Female |
Male |
Leaf a | Female |
Male |
|||
| 2-Jan | Young | 0 |
0 |
NS | Young | 0 |
0 |
NS | |
| Old | 0.8 |
0.6 |
NS | Old | 0.2 |
0 |
NS | ||
NS |
NS |
NS |
NS |
||||||
| 20-Jan | Young | 0 |
0 |
NS | Young | 0 |
0 |
NS | |
| Old | 0 |
0.1 |
NS | Old | 0.2 |
0.1 |
NS | ||
NS |
NS |
NS |
NS |
||||||
| 20-Feb | Young | 0.2 |
0.2 |
NS | Young | 0 |
0 |
NS | |
| Old | 2.2 |
1.5 |
NS | Old | 0.5 |
0.9 |
NS | ||
* |
* |
NS |
NS |
||||||
| 8-Mar | Young | 22.6 |
13.6 |
NS | Young | 0.2 |
0.2 |
NS | |
| Old | 2.9 |
2.8 |
NS | Old | 6.9 |
4.8 |
NS | ||
* |
* |
* |
* |
||||||
| NS, treatments were not significantly
differ rent, (t-test, p>0.05), * treatments were significantly different
(p<0.05) aYoung refers to terminal leaves; Old refers to lower frame leaves on plants. |
|||||||||
| Table 2. Aphid numbers at 3 days following the 1st application on a female cauliflower plants in full bloom, Yuma Mesa, Spring 2001. | ||||
March 11
(3 Days Following Application #1) |
||||
| Treatment | No. aphidsa / frame
leaf |
No. aphidsb / plant
terminal |
No. infested seed pods/ plant
|
No. aphidsb /seed
pod |
|---|---|---|---|---|
| Pirimor / Pirimor | 47.1 b |
1.4 b |
0.13 b |
12.0 d |
| Fulfill / Pirimor | 17.0 c |
90.1 b |
0.54 b |
161.2 b |
| Aphistar / Aphistar | 37.1 bc |
10.9 b |
0.16 b |
76.1 c |
| Untreated check | 98.0 a |
591.8 a |
2.00 a |
304.0 a |
| Means followed the same letter are
not significantly different ; ANOVA, protected LSD (p < 0.05) . Pre-treatment means: 91.5 aphids / frame leaf; 515.8 aphids /terminal ; 2.1 infested pods/plant. apopulation consisted primarily of Green peach aphids (>90%). b population consisted primarily of cabbage aphids (>90%). |
||||
| Table 3. Aphid numbers at 7 days following the 1st application on a female cauliflower plants in full bloom, Yuma Mesa, Spring 2001. | ||||
March 15
(7 Days Following Application #1) |
||||
| Treatment | No. aphidsa / frame
leaf |
No. aphidsb / plant
terminal |
No. infested seed pods/ plant
|
No. aphidsb /seed
pod |
|---|---|---|---|---|
| Pirimor | 67.6 b |
1.7 b |
0.22 b |
7.7 c |
| Fulfill | 45.0 b |
202.8 b |
2.50 ab |
86.3 b |
| Aphistar | 107.5 ab |
9.0 b |
0.25 b |
38.2 bc |
| Untreated check | 223.7 a |
1901.3 a |
5.35 a |
361.0 a |
| Means followed the same letter are
not significantly different ; ANOVA, protected LSD (p < 0.05) . Pre-treatment means: 91.5 aphids / frame leaf; 515.8 aphids /terminal ; 2.1 infested pods/plant. apopulation consisted primarily of green peach aphids (>90%). bpopulation consisted primarily of cabbage aphids (>90%). |
||||
| Table 4. Mean Aphid numbers on frame leaves and terminals at 7,14, 21, and 28 d following the 2nd application on female cauliflower plants in full bloom, Yuma Mesa, Spring 2001. | |||||||||
No. aphids/
frame leaf a |
No. aphids
/ terminal b |
||||||||
| Treatment | 24-Mar 7 DAT |
31-Mar 14 DAT |
7-Apr 21 DAT |
14-Apr 28 DAT |
24-Mar 7 DAT |
31-Mar 14 DAT |
7-Apr 21 DAT |
14-Apr 28 DAT |
|
| Pirimor / Pirimor | 9.1 b | 2.7 b | 3.7 b | 2.0 b | 0.6 b | 0 b | 0 b | 0 b | |
| Fulfill / Pirimor | 7.6 b | 6.2 b | 34.3 b | 1.5 b | 0.4 b | 0 b | 0 b | 0 b | |
| Aphistar / Aphistar | 11.8 b | 3.4 b | 3.3 b | 0.4 b | 0 b | 0 b | 2.5 b | 0 b | |
| Untreated check | 177.7 a | 95.7 a | 52.9 a | 20.5 a | 889.9 a | 712.0 a | 307.0 a | 205.5 a | |
| Means followed the
same letter are not significantly different ; ANOVA, protected LSD
(p < 0.05) . apopulation consisted primarily of Green peach aphids (>90%). b population consisted primarily of cabbage aphids (>90%). |
|||||||||
| Table 5. Mean Aphid numbers on frame leaves and terminals at 7,14, 21, and 28 d following the 2nd application on female cauliflower plants in full bloom, Yuma Mesa, Spring 2001. | |||||||||
No. infested
seed pods/terminal |
No. aphids
/ seed poda |
||||||||
Treatment |
24-Mar 7 DAT |
31-Mar 14 DAT |
7-Apr 21 DAT |
14-Apr 28 DAT |
24-Mar 7 DAT |
31-Mar 14 DAT |
7-Apr 21 DAT |
14-Apr 28 DAT |
|
| Pirimor / Pirimor | 0.12 b |
0 b |
0 b |
0 b |
5.7 b |
0 b |
0 b |
0 b |
|
| Fulfill / Pirimor | 0.24 b |
0 b |
0 b |
0 b |
1.5 b |
0 b |
0 b |
0 b |
|
| Aphistar / Aphistar | 0.03 b |
0.1 b |
0.1 b |
0 b |
0.3 b |
1.7 b |
2.0 b |
0 b |
|
| Untreated check | 2.0 a |
1.8 a |
1.1 a |
0.6 a |
448.5 a |
290.8 a |
234.5 a |
162.5 a |
|
| Means followed the same letter
are not significantly different ; ANOVA, protected LSD (p < 0.05)
. a population consisted primarily of cabbage aphids (>90%). |
|||||||||
| Table 6. Aphid abundance on frame leaves prior to and 11 d following the 1st application broccoli plants, pre-bloom, Yuma Ag Center, 2003 | ||||||||||
| Treatment | Mean aphids
/ leaf /plant |
|||||||||
Feb 18 (pre-spray) |
March 1
(11-DAT#1) |
|||||||||
| Rate | GPA |
CA |
TA |
Total |
GPA |
CA |
TA |
Total |
||
| Pirimor 50DF | 3 oz / acre | 2.1 |
24 |
4 |
30.1 |
1 |
1.5 |
0 |
2.5 |
|
| Fulfill 50WG | 2.75 oz | 1.2 |
4.2 |
1.2 |
6.6 |
2 |
1 |
0 |
3 |
|
| Assail 70W | 1.7 oz | 2 |
4 |
3.3 |
9.3 |
5 |
1.2 |
0.5 |
6.7 |
|
| MSR + Capture | 2 pts + 6 oz | 3 |
4.3 |
0 |
7.3 |
0 |
0 |
0 |
0 |
|
| Platinum/Capture | 8 oz + 6 oz | 1.5 |
4.1 |
0 |
5.6 |
0 |
0 |
0 |
0 |
|
| Admire/ Capture | 24 oz + 6 oz | 0 |
2.5 |
0 |
2.5 |
0 |
3.6 |
0 |
3.6 |
|
| Admire | 24 oz | 0 |
2 |
0 |
2 |
0 |
0 |
0 |
0 |
|
| Untreated | - | 4.5 |
8.2 |
0 |
12.7 |
4.5 |
13.5 |
0 |
18 |
|
| * No significant differences among treatments (AOV, LSD, P<0.05). | ||||||||||
| Table 7. Within plant distribution of aphid species in pre-blooming broccoli, Yuma Ac Center, 2003 | |||||||||
| Mean aphids / leaf | |||||||||
| Cabbage/Turnip Aphid | Green Peach Aphid | ||||||||
| Sample date | Leafa | Female | Male | Leafa | Female | Male | |||
| 28-Nov | Young | 0 |
0 |
NS |
Young | 0 |
0 |
NS |
|
| Old | 0 |
0 |
NS |
Old | 0 |
0 |
NS |
||
NS |
NS |
NS |
NS |
||||||
| 11-Dec | Young | 0 |
0 |
NS |
Young | 0 |
0 |
NS |
|
| Old | 0.1 |
0 |
NS |
Old | 0.3 |
0.4 |
NS |
||
NS |
NS |
NS |
NS |
||||||
| 30-Dec | Young | 0 |
0 |
NS |
Young | 0 |
0 |
NS |
|
| Old | 0.5 |
0.1 |
NS |
Old | 0.2 |
0.3 |
NS |
||
* |
NS |
NS |
NS |
||||||
| 14-Jan | Young | 0 |
0.1 |
NS |
Young | 0.2 |
0 |
NS |
|
| Old | 0.8 |
0.6 |
NS |
Old | 0.8 |
0.3 |
NS |
||
* |
* |
* |
NS |
||||||
| 28-Jan | Young | 0 |
5.3 |
* |
Young | 0 |
0 |
NS |
|
| Old | 1.3 |
1.8 |
NS |
Old | 1.9 |
0.5 |
NS |
||
NS |
* |
* |
NS |
||||||
| 18-Feb | Young | 3 |
3.1 |
NS |
Young | 0 |
1 |
NS |
|
| Old | 17.3 |
14 |
NS |
Old | 1 |
6 |
* |
||
* |
* |
* |
* |
||||||
| 1-Mar | Young | 20 |
6 |
* |
Young | 0 |
0 |
NS |
|
| Old | 1 |
1.5 |
NS |
Old | 3.2 |
3.3 |
NS |
||
* |
* |
* |
* |
||||||
Figure 1. Measurements of aphid infestations
on seed pod extensions on treated and untreated broccoli seed crop, YAC
2003
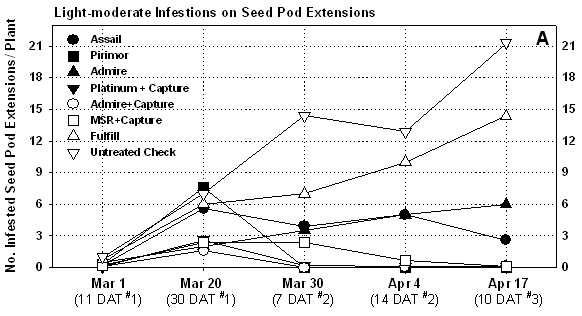
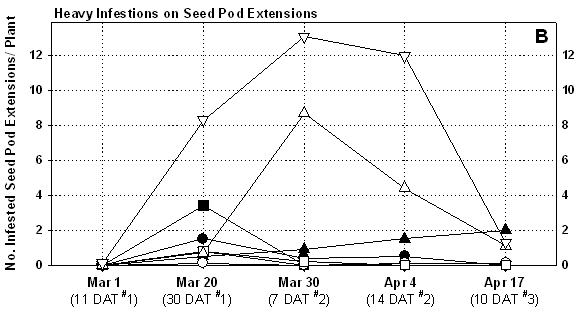
Figure 2. Estimates on plant mortality attributed to direct feeding damage by aphids on seedpod extensions, broccoli seed crop, YAC 2003
Issued in furtherance of Cooperative Extension work, acts of May 8 and June 30, 1914, in cooperation with the U.S. Department of Agriculture, James A. Christenson, Director Cooperative Extension, College of Agriculture and Life Sciences, The University of Arizona.
The University of Arizona is an equal opportunity, affirmative action institution. The University does not discriminate on the basis of race, color, religion, sex, national origin, age, disability, veteran status, or sexual orientation in its programs and activities.
Any products, services, or organizations that are
mentioned, shown, or indirectly implied in this web document do not imply
endorsement by The University of Arizona.
Information provided by:
John C. Palumbo, jpalumbo@ag.arizona.edu Research Scientist (Entomology)
Barry Tickes, btickes@ag.arizona.edu Extension Agent, Yuma County
University of Arizona, Tucson, Arizona.
Material written February 2004.
For more Arizona Production Ag Information:
Home | Cotton | Veggies| Forages | Grains | Citrus | Crop x Crop | Insects | Diseases| Weeds | Pesticides | News | Weather | Research | Photos | Contacts | General Info. | Site Map
document located at: http://cals.arizona.edu/crops/counties/yuma/farmnotes/2004/fn0204aphidseed.html
Copyright © 2001 University of Arizona,
College of Agriculture and Life Sciences
Webmaster: Al Fournier (fournier@ag.arizona.edu)